Introduction
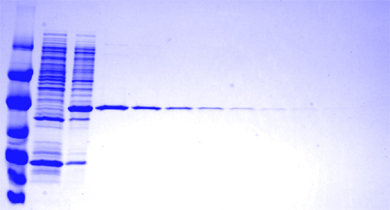
Samples were run on a gel and
coomassie brilliant blue stained to demonstrate protein purity.
Protocol
Protein
preparation
Samples were prepared as serial dilutions in PBS (phosphate
buffered saline).An equal volume of 2X denaturation gel loading
buffer was added and samples were heated to 95°C for 5 mins. A
total of 20µl of sample was loaded onto a 16% Tris-glycine gel that
was run for 60 mins at 100 volts, 120mA.
Coomassie gel and Visualisation
Gels were stained with coomassie brilliant blue, washed with
destain and visualised by UVP image analysis
Figure: Coomassie gel
| Sample |
Band on blot (left to right) |
| Protein MWT ladder |
. |
| Unpurified bacterial protein |
Note absence of 30kDa band |
| Unpurified 14-3-3 bacterial clone
fraction |
note strong ~30kDa mwt band |
| HPLC purified |
1000ng |
| HPLC purified |
500ng |
| HPLC purified |
250ng |
| HPLC purified |
125ng |
| HPLC purified |
62.5ng |
| HPLC purified |
31.25ng |
| HPLC purified |
15.62ng (weak band) |
| HPLC purified |
7.8125ng (v.v.weak) |
| HPLC purified |
3.90625ng (barely visible above
background) |