NMR and optical spectroscopy
Sophisticated expertise and spectroscopic instrumentation is hosted with the Laboratory for Molecular Structure, to support NIBSC aims for the improved characterisation of biopharmaceutical products. Consolidation of this expertise and the instrument base creates the critical mass to optimise use of these expensive facilities.
The instrumentation includes:
- A 500MHz NMR spectrometer
- Three mass spectrometers – a MALDI-TOF-TOF, a triple quadrupole and an Orbitrap instrument. The triple quadrupole and Orbitrap instruments are coupled to nanoflow HPLC systems, also see information on glycan analysis
- Circular dichroism and UV/vis optical spectroscopy
- Dynamic light scattering
This expertise enables us to undertake a wide range of projects which not only require detailed characterisation of novel products, but which are valuable for the identification of undefined products or sources of contamination. Projects include:
- Characterisation of polysaccharide and derived glycoconjugate vaccines. This class of vaccines, which include those against Haemophilus influenzae Type b, meningococcal meningitis, pneumonia and typhoid, are a critical part of childhood vaccination schedules around the world. We are using NMR and optical spectroscopy and, increasingly, chromatographic approaches to characterise them, to ensure integrity, to understand differences between products, investigate stability and determine critical quality attributes
- NMR spectroscopy provides a powerful approach to determining the structure and composition of heparin products, to identify toxic or benign contaminants and, in an on-going project, to define the species of origin.
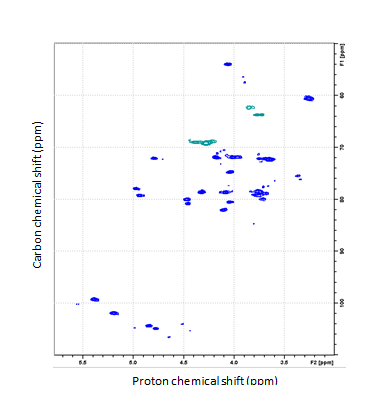
Partial 1H-13C HSQC spectrum of crude porcine heparin.
- Identification of falsified medicine, which can be achieved by defining the excipients present, by identification of contaminating compounds, or by defining the profile of process-related impurities. Complementary mass spectrometry techniques provide detailed information on protein sequence and product-related impurities.
- Multivariate statistical analysis and clustering techniques of one dimensional proton NMR data sets (“metabolonomics“) of spent growth media are being investigated as a non-invasive approach to attempt to assess where stem cells have undergone differentiation. See Imperial College.
- We undertake NMR characterisation of the structure and purity of potential reference standards for the British Pharmacopoeia
- We are developing quantitative NMR approaches to provide SI-traceable value assignments for bacterial polysaccharides used in vaccine manufacture. For these vaccines the mass of polysaccharide is the critical quality control assay, corresponding to the potency assay. We are using this approach to develop standards both for the World Health Organization (WHO) and for the United States Pharmacopeia
- Circular dichroism was used to characterise the integrity of glycoconjugate vaccines and changes in folding pattern and stability of a number of point amino acid variants of GCSF
Increasingly we are developing clustering and multivariate statistical analysis techniques to extract as much information as possible from these data-rich experimental methods. By correlating spectroscopic features with biological endpoints we hope to be able to assess the quality of complex biological products using one or more simple analytical approach.