Polysaccharide and Glycoconjugate Vaccines
The cell surface polysaccharides of many bacteria are virulence determinants, and antibodies against these components protect against disease. Initially, purified polysaccharides were developed as vaccines, and pneumococcal, typhoid and meningococcal polysaccharide vaccines continue to be used in adult populations, but they are not effective in infants. Chemical attachment of the polysaccharide to a suitable carrier protein changes the molecular mechanism and makes them effective in infants, the major risk group for many of these diseases. Conjugate vaccines against Haemophilus influenzae type b (Hib), meningitis and pneumococcal infections form part of the routine immunisation programme for infants in the UK.
Conjugation is a platform technology and many other vaccines of this type are in development, including conjugate vaccines against typhoid and paratyphoid, Group B Streptococcus and Staphylococcus aureus.
We have spent more than 30 years working on the characterisation of these products by a range of physicochemical techniques. The research work has led to the development of a range of sophisticated spectroscopic approaches to control the quality of these products.
- De novo polysaccharide structure determination (Figure 1).
> NMR methods to confirm identity of polysaccharides
> NMR methods to quantify O-acetylation
- Polysaccharide purity and identification and quantitation of impurities
- Polysaccharide stability and molecular mechanisms of degradation
> Recognition of characteristic signals highlighting product degradation
- Carrier protein structure and purity
> Mass spectrometric and optical spectroscopy approaches
- Conjugate integrity and stability
> Saccharide degradation or de-O-acetylation in the conjugate
> Thermal stability of the carrier protein in the conjugate
Structure determination: When we started this work, structures for many of the polysaccharides used were unknown. Our preliminary studies also showed errors with several reported structures. We determine the structures of these polysaccharides and obtained full NMR assignments to validate NMR identity testing methods. The figure below shows the type of spectra used in the structural analysis.
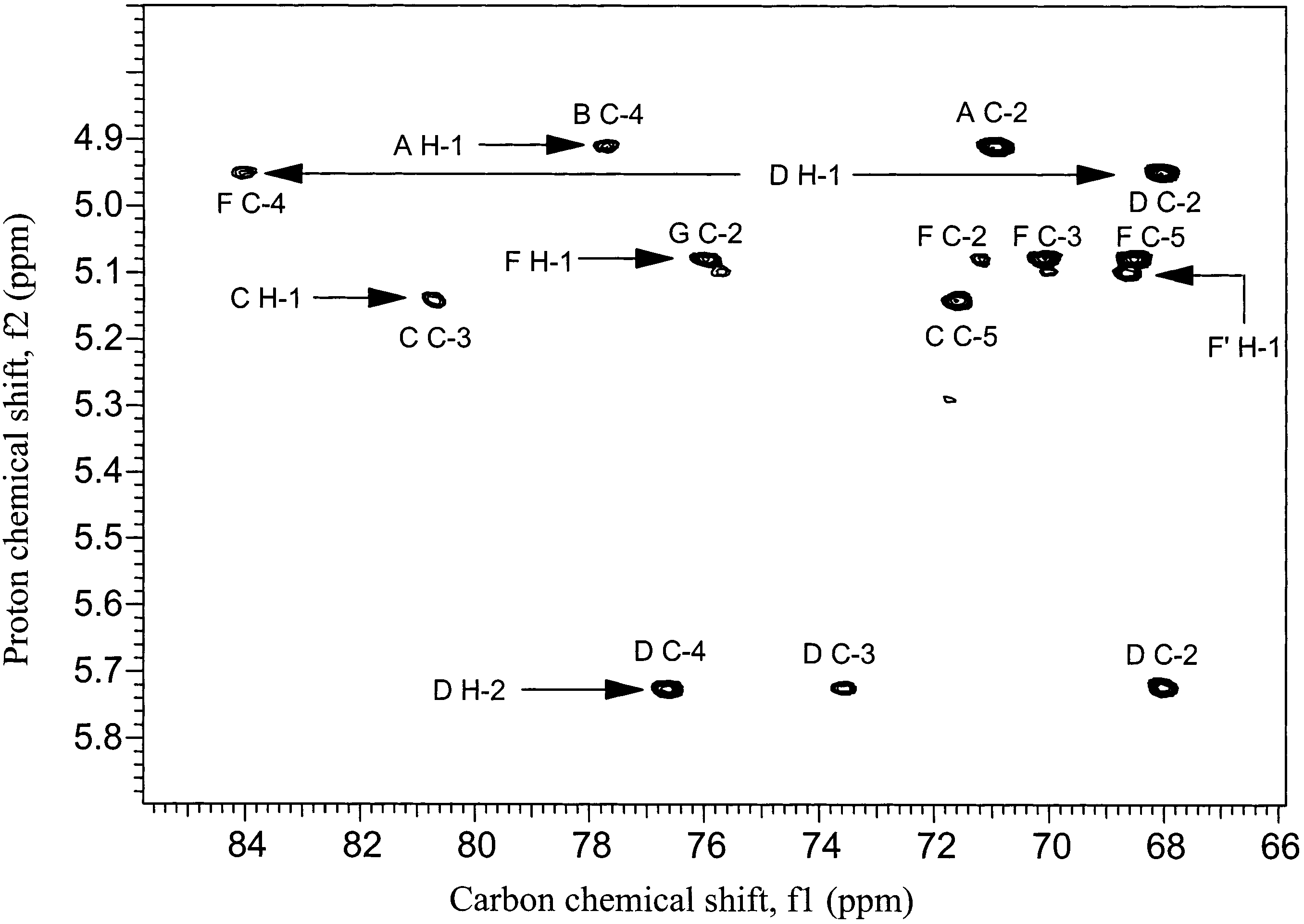 Figure 1: Partial 500MHz long-range 1H-13C correlation spectrum used to define the structure of the pneumococcal Type 17F polysaccharide, a component of pneumococcal vaccines
Figure 1: Partial 500MHz long-range 1H-13C correlation spectrum used to define the structure of the pneumococcal Type 17F polysaccharide, a component of pneumococcal vaccines
All of the major vaccine manufacturers now use NMR spectroscopy for identity testing, replacing older methods. The spectrum also contains quantitative information on impurities which may be present, such as:
- Pneumococcal C-polysaccharide, a teichoic acid
- Cetyltrimethylammonium bromide (CTAB), a reagent used in the isolation and purification of polysaccharides
- Residual solvents, such as ethanol, used in the purification of polysaccharides.
Polysaccharide degradation pathways: After heating the polysaccharides, the structural work was repeated and new groups identified. This information, combined with our knowledge of their chemistry, defines the mechanisms by they degrade. This information can help us identify degraded products.
Conjugate integrity: We developed circular dichroism (CD) methods to study the bulk conjugates. These spectra contain information on the folding of the carrier, and also information on the polysaccharide. The three traces below show CD traces of a series of developmental typhoid vaccines in which the proportion of polysaccharide to carrier protein has been varied. The spectra in Figure 2 are processed to separate the contributions from the polysaccharide and the carrier protein.
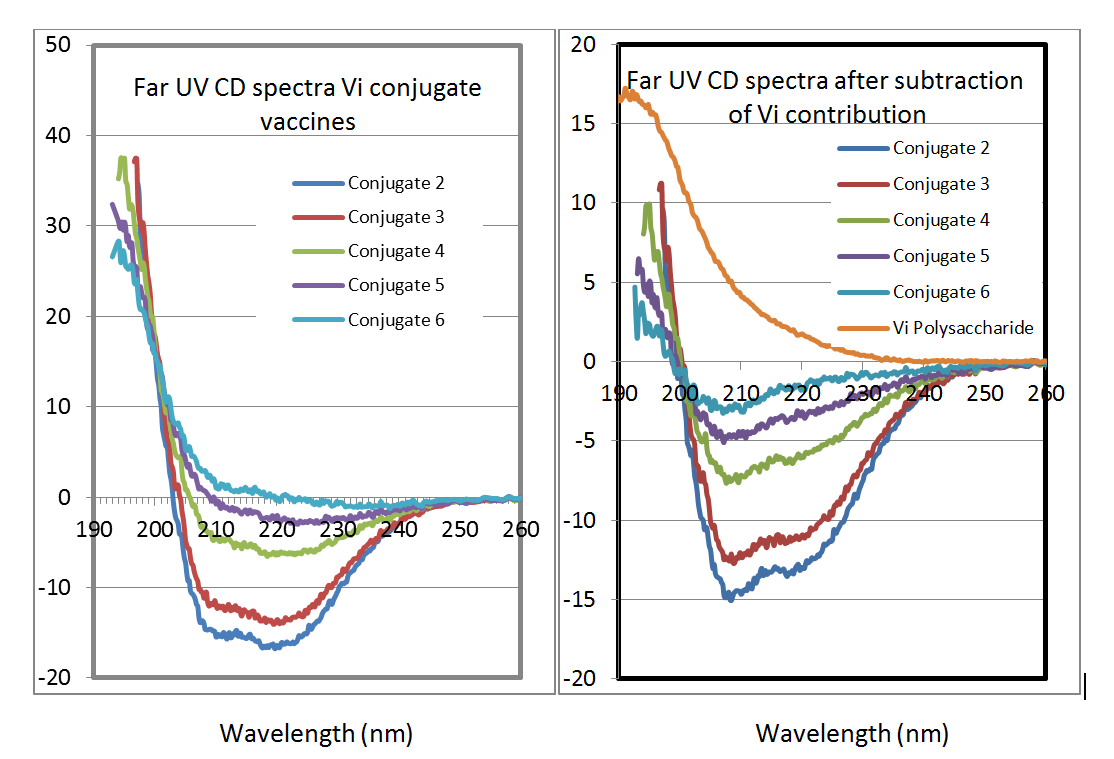 Figure 2: Far UV CD spectra of a range of typhoid Vi conjugate vaccines differing in polysaccharide: protein ratio (left) and after separation of the contributions of the carrier protein and polysaccharide (right). The spectrum of the carrier protein in the conjugate is the same as that of the carrier protein alone, showing that there has been no change in its folding as a result of the conjugation chemistry.
Figure 2: Far UV CD spectra of a range of typhoid Vi conjugate vaccines differing in polysaccharide: protein ratio (left) and after separation of the contributions of the carrier protein and polysaccharide (right). The spectrum of the carrier protein in the conjugate is the same as that of the carrier protein alone, showing that there has been no change in its folding as a result of the conjugation chemistry.
Impurities in conjugate vaccines: NMR spectra can be obtained in such a way that process impurities such as EDU can be identified and quantified.